|
While this study has the potential for registration implications for pembrolizumab in this population, the randomized phase III study is also important. It is also worth noting that this needs to be tested in a randomized setting, and that's what the KEYNOTE-676 trial is addressing. TARGETED ONCOLOGY:What are the next steps for this research?īalar:The next steps for this trial are to follow up on these patients who have achieved a CR and truly see how long and durable these responses are. Of course, these are early data, and longer-term follow-up will be necessary. About 53% of patients will have CRs lasting 9 months or longer, which is quite encouraging. Additionally, the median duration of response at this point for CR is about 12.5 months. At a median follow-up of about 14 months, results for 102 patients in cohort A showed CR rates of about 40% with pembrolizumab. Overall, this trial focused on 2 cohorts: cohort A focused on patients with carcinoma in situ with or without papillary disease, and cohort B focused on patients with papillary disease alone without carcinoma in situ. TARGETED ONCOLOGY:What findings were presented at the 2019 Genitourinary Cancers Symposium?īalar:I presented the preliminary analysis of 102 patients enrolled in cohort A of the study. In this context, pembrolizumab was tested as a means to achieve responses in the bladder and to help patients preserve their bladders. Historically, radical cystectomy has been the only curative therapy available for these patients. The reason we do this is because BCG-unresponsive disease has a natural history of progressing to muscle-invasive and eventually metastatic disease. Historically, this population is treated with radical cystectomy, meaning surgical removal of the bladder. These patients did not respond to BCG therapy. These are patients with high-risk disease who have received BCG, an intravesical immunotherapy, in the past. TARGETED ONCOLOGY:What was the rationale for the KEYNOTE-057 study?īalar:KEYNOTE-057 aimed to test pembrolizumab, a PD-1 antibody, in a specific population of patients with NMIBC. In an interview with Targeted Oncology, Balar, director of the Genitourinary Medical Oncology Program, and assistant professor in the Department of Medicine, at NYU Langone’s Perlmutter Cancer Center, highlighted the KEYNOTE-057 study findings and provided insight into the role of immunotherapy in bladder cancer. Grade 3/4 treatment-related adverse events were observed in 12.6% of patients 1 death was considered to be related to treatment. Patients received pembrolizumab at 200 mg every 3 weeks for up to 24 months. However, 80.2% of patients had a CR lasting ≥6 months.Ĭohort A was comprised of 102 patients with NMIBC who had carcinoma in situ with or without papillary disease, said Balar, who is lead author of the trial. 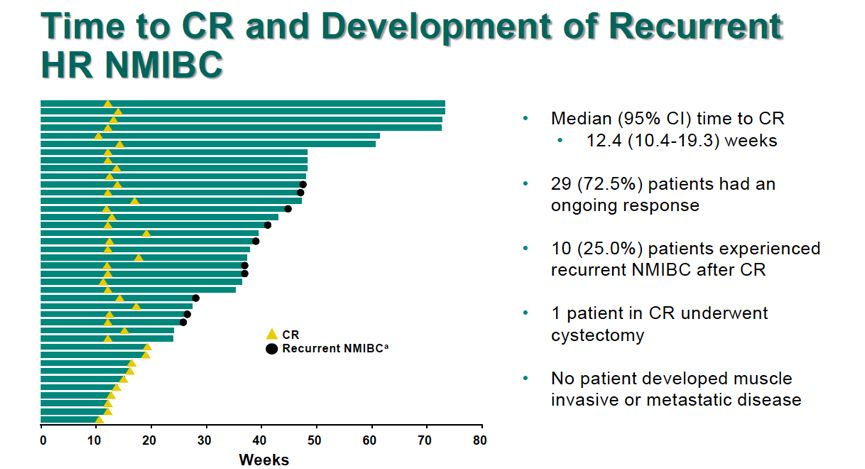
Among the 40 patients who achieved a CR at 3 months, 72.5% maintained a CR at a median follow-up of 14 months median CR duration had not been reached at the time of presentation. 
Balar, MD.ĭata for cohort A of the phase II KEYNOTE-057 study, which were presented at the 2019 Genitourinary Cancers Symposium, showed that pembrolizumab induced a 3-month complete response (CR) rate of 38.8%. Early data indicate that pembrolizumab (Keytruda) is a feasible option for patients with high-risk nonmuscleinvasive bladder cancer (NMIBC) who are unresponsive to Bacillus Calmette-Guérin (BCG), explained Arjun V.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
